Which is the correct equilibrium constant expression for the following reaction? 2C6H6(g) + 15O2(g)  12CO₂(g) + 6H₂O(g)
12CO₂(g) + 6H₂O(g)
A) 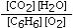
B) 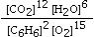
C) 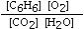
D) 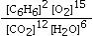
E) 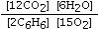
Correct Answer:
Verified
Q2: At 450°C, tert-butyl alcohol decomposes into water
Q3: Write the equilibrium constant expression for the
Q4: The reaction system POBr3(g) Q5: For the reaction 2X(g) + Y(g) Q7: Write the equilibrium constant expression for the Q9: What is defined as a fraction with Q9: Hydrogen peroxide may decompose to form water Q10: At 35ºC, the equilibrium constant for the Q11: A mixture of 0.500 mole of carbon Q66: At equilibrium, the rate of the forward![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents