How many lone pairs of electrons need to be added to complete this Lewis structure? 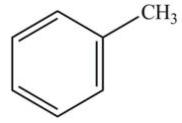
A) 5
B) 10
C) 0
D) 1
E) 6
Correct Answer:
Verified
Q1: How many lone pairs of electrons need
Q3: What does the abbreviation VSEPR stand for?
A)
Q4: What is the predicted molecular geometry of
Q6: Select the correct Lewis structure for NOCl,
Q6: Which of the following is required for
Q8: Select the correct Lewis structure for nitrogen
Q9: According to the VSEPR model, the predicted
Q53: In which one of the following species
Q60: The number of resonance structures for the
Q66: How many total resonance structures can be
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents