Consider the following equilibria: 2SO3(g) 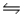
2SO2(g) + O2(g) Kc = 2.3 × 10-7
2NO3(g) 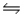
2NO2(g) + O2(g) Kc = 1.4 × 10-3
Calculate the equilibrium constant for the reaction
SO2(g) + NO3(g)
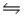
SO3(g) + NO2(g)
A) 78
B) 1.3 × 10-2
C) 1.6 × 10-4
D) 3.2 × 10-10
E) 6.1 × 103
Correct Answer:
Verified
Q35: Consider the reaction N2(g)+ O2(g) 
Q36: Consider the following reactions and their associated
Q37: At 700 K, the reaction 2SO2(g)+ O2(g)
Q38: Sodium carbonate, Na2CO3(s), can be prepared by
Q39: Hydrogen iodide decomposes according to the equation
Q41: For the following reaction at equilibrium,
Q42: Consider this reaction at equilibrium at a
Q43: For the following reaction at equilibrium
Q44: The reaction 2SO3(g) Q45: Concerning the following reaction at equilibrium: 3Fe(s)+![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents