For the following reaction at equilibrium, which gives a change that will shift the position of equilibrium to favor formation of more products? 2NOBr(g) 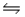
2NO(g) + Br2(g) , Hºrxn = 30 kJ/mol
A) Increase the total pressure by decreasing the volume.
B) Add more NO.
C) Remove Br2.
D) Lower the temperature.
E) Remove NOBr selectively.
Correct Answer:
Verified
Q36: Consider the following reactions and their associated
Q37: At 700 K, the reaction 2SO2(g)+ O2(g)
Q38: Sodium carbonate, Na2CO3(s), can be prepared by
Q39: Hydrogen iodide decomposes according to the equation
Q40: Consider the following equilibria: 2SO3(g) 
Q42: Consider this reaction at equilibrium at a
Q43: For the following reaction at equilibrium
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents