For the reaction CuS(s)+ H2(g) 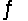 H2S(g)+ Cu(s),
H2S(g)+ Cu(s),
G°f (CuS)= -53.6 kJ/mol
G°f (H2S)= -33.6 kJ/mol
H°f (CuS)= -53.1 kJ/mol
H°f (H2S)= -20.6 kJ/mol
Calculate the value of the equilibrium constant (Kp)at 798 K and 1 atm pressure.
Correct Answer:
Verified
Q53: Using the thermodynamic data provided below, calculate
Q54: For the reaction CuS(s)+ H2(g)
Q55: Predict the normal boiling point of triethylborane
Q56: Calculate
Q57: In the gas phase, methyl isocyanate
Q59: Find the temperature at which the reaction
Q60: The standard free energy of formation of
Q63: For the reaction SbCl5(g) 
Q82: Is the reaction SiO2(s)+ Pb(s)
Q86: What is the free energy change
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents