A substance consisting of which molecule shown below should have a higher boiling point? 
A) The molecule on the left, CO 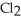 , because it has a fewer number of atoms.
, because it has a fewer number of atoms.
B) The molecule on the right, 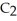
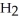 Cl, because it has a greater number of atoms.
Cl, because it has a greater number of atoms.
C) The molecule on the left, CO 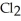 , because it is less symmetrical.
, because it is less symmetrical.
D) The molecule on the right, 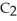
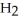 Cl, because of electronegativity considerations.
Cl, because of electronegativity considerations.
Correct Answer:
Verified
Q101: List the following bonds in order of
Q105: Which molecule is most polar?
A)S=C=S
B)O=C=O
C)O=C=S
D)These all have
Q120: Which of the following statements describes a
Q144: Which of the following molecules is the
Q145: Which of the following molecules should have
Q146: Three kids sitting equally apart around a
Q147: Would ammonia, N Q148: An individual carbon-oxygen bond is polar. Yet Q151: Which shows atoms in order of increasing Q152: Water, ![]()
![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents