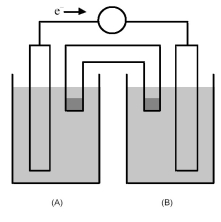
-The cell reaction 2 Fe3+(aq) + Zn(s) → Zn2+(aq) + 2 Fe2+(aq) occurs in the galvanic cell shown above.Where should the Fe3+(aq) and Fe2+(aq) be found?
A) Half-cell (A) contains Fe3+(aq) and half-cell (B) contains Fe2+(aq) .
B) Half-cell (A) contains Fe2+(aq) and half-cell (B) contains Fe3+(aq) .
C) Half-cell (A) contains Fe3+(aq) and Fe2+(aq) .
D) Half-cell (B) contains Fe3+(aq) and Fe2+(aq) .
Correct Answer:
Verified
Q113: Given the half-cell potentials below,calculate the cell
Q114: Identify the anode and cathode half-reactions and
Q115: The initial concentrations of Ag+(aq)and Cu2+(aq)are both
Q116: Fuel cells
A)produce carbon dioxide and hydrogen.
B)emit sulfur
Q117: Consider the galvanic cell shown below.
Q119: What is the shorthand notation for this
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents