Consider the formation of solid silver chloride from aqueous silver and chloride ions. Given the following table of thermodynamic data at 298 K: 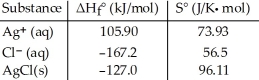 The value of K for the reaction at 25 °C is ________.
The value of K for the reaction at 25 °C is ________.
A) 1.9 × 10-10
B) 810
C) 1.8 × 104
D) 3.7 × 1010
E) 5.3 × 109
Correct Answer:
Verified
Q92: Consider the reaction: FeO (s)+ Fe (s)+
Q93: The signs of ΔH° and ΔS° must
Q94: ΔS is negative for the reaction _.
A)Sr(NO3)2
Q95: The standard Gibbs free energy of formation
Q96: Consider the reaction between ammonia and hydrochloric
Q98: Which of the following has the largest
Q99: The value of ΔG° at 181.0 °C
Q100: What is the equilibrium constant for a
Q101: At what temperature (in K)will a reaction
Q102: What is the equilibrium constant for a
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents