Consider the boiling points of the following compounds and their solubilities in room-temperature water. Why does the solubilities in water go down as the boiling points of these alcohols go up. 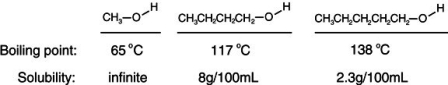
A) Larger molecules are less attracted to one another by induced dipole-induced dipole as well as by dipole-dipole and dipole-induced dipole attractions.
B) As the boiling increases, it is more difficult to keep the alcohol from evaporating out of solution.
C) As the boiling point increases, the size of the alcohol molecules decreases.
D) Larger molecules are more attracted to one another by induced dipole-induced dipole as well as by dipole-dipole and dipole-induced dipole attractions.
Correct Answer:
Verified
Q26: In a solution of 77 percent nitrogen
Q29: Which of the following statements describes a
Q38: Dipole-induced dipole forces of attraction exist between
Q47: The boiling point of 1,4-butanediol is 230°C.
Q48: What is molarity?
A)the number of moles of
Q51: Why are the melting temperatures of most
Q52: A 1 molar solution of sugar water
Q59: A gemstone is an example of a
Q126: Which of the following would have the
Q142: Why is the surface area of a
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents