The boiling point of 1,4-butanediol is 230°C. Would you expect this compound to be soluble or insoluble in room-temperature water? 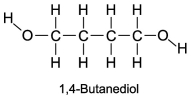
A) Since there are no polar areas on this molecule, it is insoluble in water at room temperature.
B) A high boiling point means that the substance interacts with itself quite strongly. Therefore this molecule is not soluble in water.
C) Since there are polar areas on this molecule, it is insoluble in water at room temperature.
D) Water would be attracted to both ends of 1,4 butanediol, and it is infinitely soluble in water.
Correct Answer:
Verified
Q26: In a solution of 77 percent nitrogen
Q27: Which of the following describes the term
Q28: Which of the following material phases cannot
Q43: Consider the boiling points of the following
Q48: What is molarity?
A)the number of moles of
Q51: Why are the melting temperatures of most
Q51: Two chemical structures are shown, one of
Q52: A 1 molar solution of sugar water
Q58: What statement best describes a mole?
A)a little
Q59: A gemstone is an example of a
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents