Gas A is composed of diatomic molecules (two atoms per molecule) of a pure element. Gas B is composed of triatomic molecules (three atoms per molecule) of another pure element. A volume of gas B is found to be three times more massive than an equal volume of gas A. How does the mass of an atom of gas B compare with the mass of an atom of gas A? 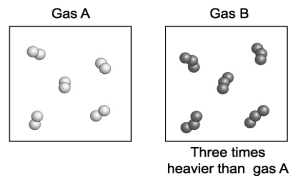
A) An atom of gas B is six times the mass of an atom of gas A.
B) An atom of gas B is three times the mass of an atom of gas A.
C) An atom of gas B is two times the mass of an an atom of gas A.
D) An atom of gas B is equivalent to the mass of an atom of gas A.
Correct Answer:
Verified
Q23: If the relative mass of a hydrogen
Q29: What is the formula mass of a
Q31: Why is it important for a chemist
Q36: Which equation best describes the reaction represented
Q38: What is the formula mass of sulfur
Q39: The reactants shown schematically below represent iron
Q41: The following diagrams depict the reaction between
Q45: What is the mass of one mole
Q48: How many grams of water can be
Q50: How many grams of water, H2O, can
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents