The following diagrams depict the reaction between gaseous chlorine, 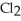 , and gaseous hydrogen,
, and gaseous hydrogen, 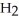 , to form gaseous hydrogen chloride, HCl. What should be drawn in the empty box and how many grams of products are formed?
, to form gaseous hydrogen chloride, HCl. What should be drawn in the empty box and how many grams of products are formed? 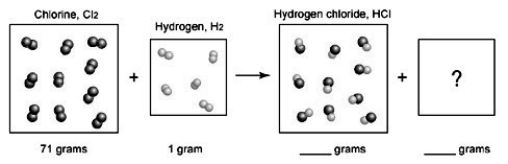
A) five 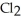 molecules in empty box; 37 grams of HCl + 36 grams of
molecules in empty box; 37 grams of HCl + 36 grams of 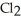
B) five 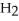 molecules in empty box; 71 grams of HCl + 1 gram of
molecules in empty box; 71 grams of HCl + 1 gram of 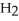
C) five HCl molecules in empty box; 50 grams of HCl + 22 grams of HCl
D) five 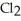 molecules in empty box; 71 grams of HCl + 1 gram of
molecules in empty box; 71 grams of HCl + 1 gram of 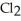
Correct Answer:
Verified
Q23: If the relative mass of a hydrogen
Q24: You are given two samples of elements,and
Q29: What is the formula mass of a
Q36: Which equation best describes the reaction represented
Q38: What is the formula mass of sulfur
Q39: The reactants shown schematically below represent iron
Q40: Gas A is composed of diatomic molecules
Q45: What is the mass of one mole
Q48: How many grams of water can be
Q50: How many grams of water, H2O, can
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents