How many moles of water, 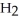 O, are produced from the reaction of 16 grams methane, C
O, are produced from the reaction of 16 grams methane, C 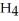 , with an unlimited supply of oxygen,
, with an unlimited supply of oxygen, 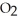 . How many grams of
. How many grams of 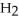 O is this? C
O is this? C 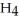 + 2
+ 2 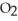 → C
→ C 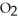 + 2
+ 2 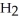 O
O
A) 0.889 mole, which is 16 grams
B) 2.0 moles of water, which is 32 grams
C) 2.0 moles of water, which is 36 grams
D) 1.0 mole of water, which is 18 grams
Correct Answer:
Verified
Q81: In the laboratory, endothermic reactions are usually
Q86: How much energy, in kilojoules, is released
Q87: What is an exothermic reaction?
A)It is a
Q90: Use the bond energies below to determine
Q101: Which is higher in an endothermic reaction:
Q104: Bond energies increase in going from C-N
Q110: Are the chemical reactions that take place
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents