Use the bond energies below to determine whether the following reaction is exothermic or endothermic: 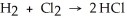 H-H (bond energy: 436 kJ/mol) Cl-Cl (bond energy: 243 kJ/mol)
H-H (bond energy: 436 kJ/mol) Cl-Cl (bond energy: 243 kJ/mol)
H-Cl (bond energy: 431 kJ/mol)
A) Exothermic with more than 50 kJ of energy released.
B) Endothermic with more than 50 kJ of energy absorbed.
C) Exothermic with less than 50 kJ of energy released.
D) Endothermic with less than 50 kJ of energy absorbed.
Correct Answer:
Verified
Q82: In a chemical reaction, the bonds being
Q86: How much energy, in kilojoules, is released
Q87: How many moles of water, 
Q87: What is an exothermic reaction?
A)It is a
Q98: What is an endothermic reaction?
A)It is a
Q101: Which is higher in an endothermic reaction:
Q104: Bond energies increase in going from C-N
Q109: If it takes energy to break bonds
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents