The chemical formula CuBr2 indicates that this compound is composed of 1 gram of copper and 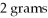
of bromine.
Correct Answer:
Verified
Q21: The molecular formula is equal to the
Q23: C2H3O2 could be an empirical formula.
Q26: Water is 11.2% hydrogen by mass.
Q27: One mole of lead(II)nitrate contains six moles
Q28: The correct formula for calculating mass percent
Q29: A molecule that has an empirical formula
Q31: Which of the following statements about the
Q37: The molar mass of a compound serves
Q38: What is the correct value for Avogadro's
Q40: The empirical formula mass is 18.0 and
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents