How many mL of 0.218 M sodium sulfate react with exactly 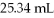
Of 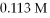
BaC 
Given the reaction:
Ba 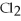
(aq) + N 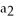
S 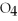
(aq) → BaS 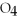
(s) + 2NaCl (aq)
A) 13.1
B) 5.52
C) 24.6
D) 2.86
E) none of the above
Correct Answer:
Verified
Q82: Which one of the following aqueous solutions
Q83: How many milliliters of 0.755 M H2SO4
Q85: In comparing a 0.25 molality aqueous NaCl
Q87: After you have completed the task of
Q89: What is the final concentration of a
Q89: What are the ion concentrations in a
Q90: How much water would you ADD to
Q91: What volume of 9.00 M nitric acid
Q95: A 0.15 M solution of BaCl2 contains:
A)0.15
Q99: A 90.0 g sample of NaOH is
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents