What is the final concentration of a solution prepared by diluting 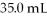
Of 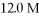
HCl to a final volume of 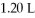
?
A) 0.504 M
B) 3.50 M
C) 0.420 M
D) 0.350 M
E) none of the above
Correct Answer:
Verified
Q82: Which one of the following aqueous solutions
Q84: Which solution below contains the highest total
Q84: What is the concentration of sodium chloride
Q85: How many grams of barium sulfate are
Q85: In comparing a 0.25 molality aqueous NaCl
Q90: How much water would you ADD to
Q91: What volume of 9.00 M nitric acid
Q92: If you add 4.00 mL of pure
Q94: How many mL of 0.218 M sodium
Q98: Oftentimes solubility of a compound limits the
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents