Calculate the mass percent of a NaCl solution prepared by mixing 47.0 g NaCl with 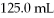
Of pure water.
A) 27.3
B) 72.3
C) 47.0
D) 37.6
E) none of the above
Correct Answer:
Verified
Q102: An osmosis cell is constructed of a
Q104: What is the final concentration (M)of a
Q105: How many moles of KCl are present
Q106: What is the molality of a solution
Q107: Calculate the molarity of a KCl solution
Q111: How many grams of KCl are present
Q115: Which of the following aqueous solutions has
Q116: What volume (L)of 2.00 M KCl solution
Q118: Why is it NOT a good idea
Q120: Solution A has a concentration of 0.10
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents