How many moles of KCl are present in 95.3 mL of 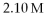
KCl?
A) 0.0500
B) 5.00
C) 20.0
D) 0.200
E) none of the above
Correct Answer:
Verified
Q100: What molarity should the stock solution be
Q101: What is the change in the boiling
Q103: What is the change in the freezing
Q104: What is the final concentration (M)of a
Q106: Determine the volume (liters)of 0.500M NaOH solution
Q106: What is the molality of a solution
Q107: Calculate the molarity of a KCl solution
Q109: Calculate the mass percent of a NaCl
Q116: What volume (L)of 2.00 M KCl solution
Q118: Why is it NOT a good idea
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents