Multiple Choice
What is the pH of a solution that has a H⁺ concentration equal to 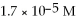
?
A) 4.77
B) 5.20
C) 0.22
D) 10.20
E) none of the above
Correct Answer:
Verified
Related Questions
Q81: In order for a solution to be
Q82: What is the concentration of hydronium ions
Q86: What is the concentration of H⁺ in
Q88: The pH of a solution is 5.00.Which
Q90: Substances that can act both as an
Q90: What is the concentration of the hydronium
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents