Consider a 1.0 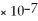
M HCl solution and a 1.0 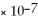
M NaOH solution.Which statement below is true?
A) The HCl solution is acidic.
B) The NaOH solution is basic.
C) Only the HCl solution is neutral.
D) Only the NaOH solution is neutral.
E) Both of these solutions are neutral.
Correct Answer:
Verified
Q81: In order for a solution to be
Q87: Which solution below has the highest concentration
Q88: What is the pH of a solution
Q88: The pH of a solution is 5.00.Which
Q90: What is the concentration of the hydronium
Q90: Substances that can act both as an
Q94: What is the concentration of H⁺ in
Q95: What is the concentration of the hydroxide
Q96: What is the value of the ion
Q100: Ammonia (NH3)ionizes in water to form a
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents