Refer to the figure below. 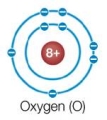 An oxygen atom by itself is very reactive, whereas an oxygen atom that has combined with two hydrogen atoms to form a water molecule is very stable.What explains this observation?
An oxygen atom by itself is very reactive, whereas an oxygen atom that has combined with two hydrogen atoms to form a water molecule is very stable.What explains this observation?
A) Oxygen starts out with a total of eight electrons and becomes more stable when it adds more electrons.
B) The eight protons in the oxygen nucleus need additional electrons from hydrogen atoms to help neutralize their charge.
C) The two electrons in the innermost shell of an oxygen atom need two additional electrons to balance out.
D) The mass provided by the two hydrogen atoms adds to the mass of the oxygen atom, which increases the stability of the entire structure.
E) An oxygen atom only has six electrons in its valence shell and needs two electrons from hydrogen to fill this shell.
Correct Answer:
Verified
Q138: Refer to the figure below. 
Q139: Refer to the figure below showing various
Q140: Refer to the table below. 
Q141: Scientists have used glucose labeled with carbon-14
Q142: Which is not an example of a
Q144: A single carbon atom can bond a
Q145: What type of chemical bond connects the
Q146: The five statements below describe properties of
Q147: What type of chemical bond connects the
Q148: Covalent bonds
A) include polar but not nonpolar
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents