Refer to the figure below. 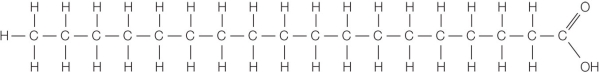 The figure shows a molecule found in biological samples.What interactions will occur if 1 part of this compound is mixed with 20 parts of water, and why?
The figure shows a molecule found in biological samples.What interactions will occur if 1 part of this compound is mixed with 20 parts of water, and why?
A) Hydrophobic interactions will cause these molecules to disperse evenly between the water molecules.
B) Hydrophilic interactions will cause these molecules to disperse evenly between the water molecules.
C) Hydrophobic interactions between these molecules and water will cause all of the molecules to form a separate layer.
D) Both hydrophobic and hydrophilic interactions between these molecules and water will cause groups of the molecules to form small spherical bunches dispersed in the water.
E) Both hydrophobic and hydrophilic interactions between these molecules and water will cause single molecules to disperse evenly between the water molecules.
Correct Answer:
Verified
Q159: Oxygen (O) has an atomic number of
Q160: Which statement is true?
A) A single covalent
Q161: The opposite of hydrophobic is
A) hydrophilic.
B) hygroscopic.
C)
Q162: The figures below have all been used
Q163: Which is an instance of van der
Q165: Refer to the figure below. 
Q166: Which statement is false?
A) Covalent bonds can
Q167: Which experimental procedure could be used to
Q168: Refer to the figure below. 
Q169: Refer to the figure below. 
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents