What is the excess reactant for the reaction below given that you start with 10.0 grams of Al and 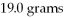
Of O2?
Reaction: 4Al + 3O2 → 2Al2O3
A) Al
B) O2
C) Al2O3
D) both Al and O2
E) not enough information
Correct Answer:
Verified
Q62: Which ingredient is the limiting reactant if
Q74: Consider the following generic chemical equation: 2A
Q75: If the theoretical yield of a reaction
Q77: The theoretical yield of a reaction is
Q84: Which is the limiting reactant in the
Q86: How many moles of NH3 can be
Q87: What is the limiting reactant for the
Q89: If 16.0 grams of aluminum oxide were
Q98: The reaction for the oxidation of NH3
Q99: How many moles of calcium chloride are
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents