What is the limiting reactant for the following reaction given we have 2.6 moles of HCl and 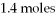
Of Ca(OH) 2?
Reaction: 2HCl + Ca(OH) 2 → 2H2O + CaCl2
A) Ca(OH) 2
B) HCl
C) H2O
D) CaCl2
E) not enough information
Correct Answer:
Verified
Q82: What is the excess reactant for the
Q84: Which is the limiting reactant in the
Q86: How many moles of NH3 can be
Q88: A 24.0 g sample of nitrogen gas
Q89: If 16.0 grams of aluminum oxide were
Q90: Calculate the maximum number of grams of
Q90: What is the limiting reactant for the
Q96: What is the limiting reactant for the
Q97: How many grams of the excess reactant
Q98: The reaction for the oxidation of NH3
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents