What is the pressure of a 1.0 L flask containing 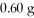
Of He at 25°C? (R= 0.0821 L atm/ mol K)
A) 98 atm
B) 3.7 atm
C) 7.3 atm
D) 15 atm
E) none of the above
Correct Answer:
Verified
Q104: Hydrogen gas produced in the laboratory by
Q105: How many liters of O2 (g)are needed
Q107: What is the volume (in liters)of 1.00
Q113: At STP,12.69 g of a noble gas
Q113: What is the final volume of a
Q114: The vapor pressure of water at 20.0°C
Q115: Ammonia gas decomposes according to the equation:
Q115: What is the molecular weight of a
Q116: What is the final pressure of a
Q121: What is the volume of 28.0 g
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents