Automobile air bags inflate during a crash or sudden stop by the rapid generation of nitrogen gas from sodium azide. 2NaN3(s)  2Na(s)
2Na(s) 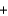 3N2(g)
3N2(g)
How many moles of sodium azide are needed to produce sufficient nitrogen to fill a 50.0 L air bag to a pressure of 1.25 atm at 25°C?
A) 2.56 mol
B) 3.83 mol
C) 5.31 mol
D) 1.70 mol
E) 4.92 mol
Correct Answer:
Verified
Q50: The pressure gauge on a cylinder
Q51: What is the temperature if 3.0
Q52: Which of the gas samples shown below
Q53: Considering that PV = nRT, which
Q54: What is the pressure of a
Q56: In an experiment, 25.0 mL of
Q57: A 22.4-L sealed chamber at 273 K
Q58: In the presence of a catalyst, potassium
Q59: Which of the gas samples shown below
Q60: Which of the following reactions will result
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents