Which is the limiting reactant in the following reaction given that you start with 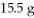
Of Na2S and 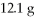
CuSO4?
Reaction: Na2S + CuSO4 → Na2SO4 + CuS
A) Na2S
B) CuSO4
C) Na2SO4
D) CuS
E) not enough information
Correct Answer:
Verified
Q83: How many grams of water are theoretically
Q84: In order to determine the limiting reactant
Q88: How many grams of aluminum oxide are
Q90: What is the limiting reactant for the
Q91: How many moles of aluminum oxide are
Q92: A sample of 8.5 g NH3 on
Q97: How many grams of the excess reactant
Q98: What is the excess reactant for the
Q99: Which is the excess reactant in the
Q101: Starting with 156 g ![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents