Which of the following reactions will result in a negligible change in pressure?
A) Na(s) 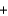 Cl2(g)
Cl2(g)  NaCl(s)
NaCl(s)
B) CO(g) 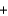 H2O(l )
H2O(l )  CO2(g)
CO2(g) 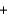 H2(g)
H2(g)
C) CH4(g) 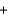 H2O(l )
H2O(l )  CO(g)
CO(g) 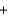 3H2(g)
3H2(g)
D) BrNO2(g)  NO(g)
NO(g)  NO2(g)
NO2(g) 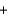 BrNO(g)
BrNO(g)
E) 2Na(s) 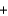 2H2O(g) 2NaOH(s)
2H2O(g) 2NaOH(s) 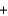 H2(g)
H2(g)
Correct Answer:
Verified
Q44: The pressure gauge on a 100-L cylinder
Q45: How many moles of propane are
Q46: What volume is occupied by 1.00
Q47: Which one of these samples contains
Q48: Calcium carbonate (100.1 g/mol) decomposes according to
Q50: The pressure gauge on a cylinder
Q51: What is the temperature if 3.0
Q52: Which of the gas samples shown below
Q53: Considering that PV = nRT, which
Q54: What is the pressure of a
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents