Passage
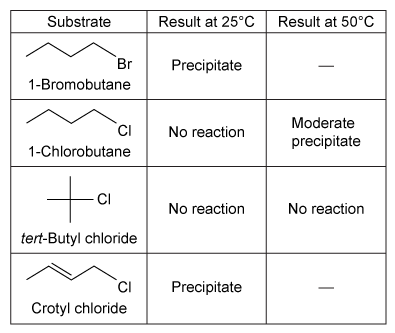
A group of students studied the reactivity of four alkyl halides (R−X) in two nucleophilic substitution reactions. Nucleophilic substitution reactions can go through one of two mechanisms. The SN2 reaction requires substitution of a leaving group with a nucleophile in a single step (Reaction 1) , and the SN1 reaction requires elimination of the leaving group and formation of an intermediate before nucleophilic substitution can occur, resulting in a two-step process (Reaction 2) .R−X + NaI → R−I + NaX↓Reaction 1R−X + AgNO3 → R+ + AgX↓ + HOCH2CH3 → R−OCH2CH3 + HNO3Reaction 2Experiment 1The students performed the SN2 reaction (Reaction 1) by the addition of a few drops of each alkyl halide into a separate test tube containing 1 mL of 15% solution of NaI in acetone at 25°C. After mixing, they monitored the reaction for the formation of a precipitate (sodium halide salt) . If no precipitate formed after 5 minutes, they raised the temperature of the reaction to 50°C and again checked for the formation of a precipitate. Formation of a precipitate indicated the substrate underwent nucleophilic substitution. The results observed for the Finkelstein reaction are shown in Table 1.Table 1 Results of the SN2 Reaction
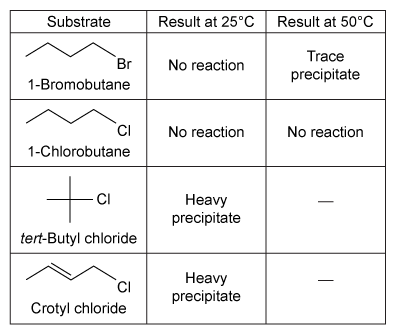
 Experiment 2The students repeated the procedure followed in Experiment 1 except they added a few drops of each alkyl halide into a separate test tube containing 1 mL of 1% solution of AgNO3 in ethanol at 25°C. The results observed for this reaction are shown in Table 2.Table 2 Results of the SN1 Reaction
Experiment 2The students repeated the procedure followed in Experiment 1 except they added a few drops of each alkyl halide into a separate test tube containing 1 mL of 1% solution of AgNO3 in ethanol at 25°C. The results observed for this reaction are shown in Table 2.Table 2 Results of the SN1 Reaction
- Compounds 1 and 2 are both intermediates that could form by a reaction with AgNO3 in ethanol, but Compound 1 is less stable than Compound 2 because:
Compounds 1 and 2 are both intermediates that could form by a reaction with AgNO3 in ethanol, but Compound 1 is less stable than Compound 2 because:
A) Compound 1 contains an electron donating group that destabilizes the carbocation.
B) there is an inductive effect from the fluorine atoms that destabilizes the carbocation.
C) the methyl groups on the structure withdraw electrons from the positively charged carbon.
D) the electronegative fluorine atoms cause the carbocation to rearrange into a secondary carbocation.
Correct Answer:
Verified
Q78: The (R)-enantiomer of the antiasthma drug albuterol
Q79: A solution of Compound 1, shown below,
Q80: Passage
Fluorescent amino acids are useful for labeling
Q81: Passage
Electrospray ionization mass spectrometry (ESI-MS) employs a
Q82: Passage
Electrospray ionization mass spectrometry (ESI-MS) employs a
Q84: Passage
A group of students studied the reactivity
Q85: Which of the following statements accurately describes
Q86: Passage
A group of students studied the reactivity
Q87: Passage
A group of students studied the reactivity
Q88: What structure is equivalent to Compound 1
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents